- 2021 IDSA/ACG Updated Guidelines for Managing Clostridioides difficile Infection
- 2021 ACG Managing Acute Upper Gastrointestinal Bleeding from Ulcers
- 2021 ACG Practical Approach to Identifying and Managing Idiosyncratic Drug-Induced Liver Injury
- 2021 AHA/ASA Guidelines for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack
- 2021 AAD Actinic Keratosis Clinical Practice Guidelines
- 2021 NRN Diagnosis and management of Guillain–Barré syndrome
- 2021 WSES-AAST Emergency Management of Inflammatory Bowel Disease
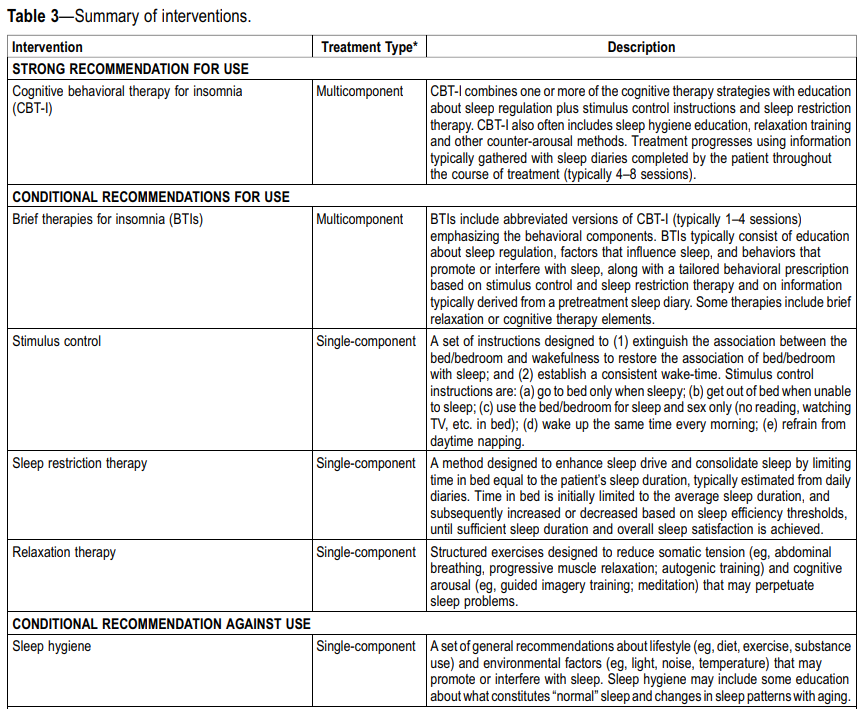
- 2021 AASM Behavioral and Psychological Treatments for Chronic Insomnia Disorder
- 2021 ASPN Cancer-Associated Pain Clinical Practice Guidelines
- 2021 CDC Sexually Transmitted Infections Clinical Practice Guidelines
For patients with nonfulminant CDI, the IDSA guidelines favor use of fidaxomicin over vancomycin.
In addition, for patients with recurrent CDI and prior episode in the last six months, the IDSA guidelines favor use of adjunctive bezlotoxumab (with a standard antibiotic regimen).
For patients with recurrent CDI who are not fecal microbiota transplantation candidates, the ACG guidelines suggest use of suppressive oral vancomycin following completion of treatment.
Endoscopic therapy is recommended in patients with UGIB due to ulcers with active spurting, oozing, and nonbleeding visible vessels. Erythromycin infusion (250mg) is suggested before endoscopy, and endoscopy is suggested within 24 hours after presentation.
After endoscopic hemostasis, high-dose proton pump inhibitor therapy is recommended continuously or intermittently for 3 days, followed by twice-daily oral proton pump inhibitor for the first 2 weeks of therapy after endoscopy. Repeat endoscopy is suggested for recurrent bleeding, and if endoscopic therapy fails, transcatheter embolization is suggested.
The hallmark of DILI treatment is withdrawal of the offending medication especially when there is evidence of liver dysfunction.
There are no definitive therapies for idiosyncratic DILI with or without acute liver failure (ALF), N-acetylcysteine (NAC) treatment is suggested in early stage ALF per good safety profile and some evidence for efficacy in early coma stage.
There are no well-conducted studies to either recommend or refute corticosteroid therapy in patients with DILI.
For symptomatic pruritis, most clinicians use antihistamines (diphenhydramine or hydroxyzine).
Lifestyle factors, including healthy diet and physical activity, are important for preventing a second stroke. Low-salt and Mediterranean diets are recommended for stroke risk reduction.
Antithrombotic therapy, including antiplatelet or anticoagulant agents, is recommended for nearly all patients without contraindications. With very few exceptions, the combination of antiplatelets and anticoagulation is typically not indicated for secondary stroke prevention. Dual antiplatelet therapy is not recommended long term, and short term, dual antiplatelet therapy is recommended only in very specific patients, including those with early arriving minor stroke and high-risk transient ischemic attack or severe symptomatic intracranial stenosis.
Atrial fibrillation remains a common and high-risk condition for second ischemic stroke. Anticoagulation is usually recommended if the patient has no contraindications.
Patients with severe intracranial stenosis in the vascular territory of ischemic stroke or transient ischemic attack should not receive angioplasty and stenting as a first-line therapy for preventing recurrence. Aggressive medical management of risk factors and short-term dual antiplatelet therapy are preferred.
Patients with embolic stroke of uncertain source should not be treated empirically with anticoagulants or ticagrelor because it was found to be of no benefit.
For hyperlipidemia treatment, target LDL-C <70 mg/dL was superior to a target of 90 to 110 mg/dL for preventing major cardiovascular events. High-risk patients with ASCVD should receive high-intensity statin therapy and that if LDL-C remains ≥70 mg/dL (≥1.8 mmol/L) on maximally tolerated statin therapy, it may be reasonable to add ezetimibe and then a PCSK-9 inhibitor if necessary and if patients are deemed to be at very high risk.
Treatment of AK can be field directed or lesion directed. Field directed treatments, such as topical agents or PDT, can be used to manage multiple AKs and keratinocyte changes in a contiguous area and may provide benefits in reducing the risk of developing new AKs, limiting AK recurrence, and mitigating subclinical damage.
Topical agent options include: 5-fluorouracil, imiquimod (strong recommendations) and diclofenac (conditional recommendation).
Lesion-directed treatments are used to manage few or isolated AKs.
UV protection with sunscreen is strongly recommended for remission and reduction of AKs.

For patients with severe Guillain-Barré syndrome (GBS) whose symptoms worsen or fail to improve after a course of intravenous immune globulin (IVIG), a repeat course has sometimes been given, despite uncertain benefit. In a randomized trial of 93 patients with GBS and a poor predicted outcome, those assigned to a second course of IVIG (given two to four days after completion of the first course) had similar disability but more adverse effects, including thromboembolic complications, than those who were assigned to placebo. Based on these data, we suggest against retreating with a second course of IVIG for patients with GBS.
Inflammatory bowel disease (IBD) encompasses a group of chronic inflammatory disorders comprising most commonly of ulcerative colitis (UC) and Crohn’s disease (CD).
Laboratory tests are strongly recommended for assessing CD or UC, including: blood count, electrolytes, liver enzymes, inflammatory biomarkers such as erythrocyte sedimentation rate (ESR) and C Reactive Protein (CRP), serum albumin and pre-albumin (to assess nutritional status and degree of inflammation).
If there are radiological percutaneous drainage of intra-abdominal abscesses > 3 cm related to Crohn’s disease, administer early empiric administration of antibiotics to adapt these as soon as possible to microbiological cultures results.
The initial medical treatment for severe active UC is intravenous corticosteroids, then assessed by the third day.
In non-responder hemodynamically stable patients, medical rescue therapy including infliximab in combination with a thiopurine, or ciclosporin should be considered in a multidisciplinary approach. Infliximab should be considered if anti-inflammatory therapy for penetrating ileocecal Crohn’s disease.
Preoperative treatments with immunomodulators associated with anti-TNF-α agents and steroids are risk factors for intra-abdominal sepsis in patients requiring emergency resectional surgery.
In complex perianal fistulizing disease infliximab or adalimumab can be used as first line therapy in combination with azathioprine following adequate surgical drainage if indicated. A combination of ciprofloxacin and anti-TNF improves short term outcomes.
Opioids for cancer pain – Opioids should be considered for moderate to severe cancer-related pain. Opioid agent selection should be individualized to account for the variance in pain presentations and co-existing medical comorbidities.
Methadone should be considered when other opioids are ineffective, or additional NMDA or serotonin receptor modulation is desired. Dosing initiation is dependent on opioid tolerance with low introductory doses for naïve patients. For opioid tolerant patients a conservative approach is recommended starting at 75–90% less than the calculated equianalgesic dose using 1:15 to 1:20 conversion factor.
Ketamine therapy for cancer pain should be considered on a case-by-case basis for refractory neuropathic, bone, and mucositis-related pain.
Intrathecal drug delivery using an implantable pump should be strongly considered in patients with cancer-related pain that is not responding to conventional medical management. Trialing before intrathecal pump implantation for cancer-related pain should be optional and at the discretion of the physician and patient.
Generally speaking, this link provides a comprehensive amount of knowledge on the management of STDs, STIs, vaccinations, prevention, screening/testing, diagnostic, special populations, allergy alternatives, and more. This brief segment only covers the drug therapy.
HIV
ART should be initiated as soon as possible for all persons with HIV infection regardless of CD4+ T-cell count, both for individual health and to prevent HIV transmission
Chancroid
Azithromycin 1 g orally in a single dose or
Ceftriaxone 250 mg IM in a single dose or
Ciprofloxacin 500 mg orally 2 times/day for 3 days or
Erythromycin base 500 mg orally 3 times/day for 7 days
Genital Herpes (First episode)
Acyclovir† 400 mg orally 3 times/day for 7–10 days or
Famciclovir 250 mg orally 3 times/day for 7–10 days or
Valacyclovir 1 g orally 2 times/day for 7–10 days
- Treatment can be extended if healing is incomplete after 10 days of therapy.
Genital Herpes (Suppression)
Acyclovir 400 mg orally 2 times/day or
Valacyclovir 500 mg orally once a day or
Valacyclovir 1 g orally once a day or
Famciclovir 250 mg orally 2 times/day
Genital Herpes (Recurrent episode)
Acyclovir 800 mg orally 2 times/day for 5 days or
Acyclovir 800 mg orally 3 times/day for 2 days or
Famciclovir 1 g orally 2 times/day for 1 day or
Famciclovir 500 mg orally once, followed by 250 mg 2 times/day for 2 days or
Famciclovir 125 mg orally 2 times/day for 5 days or
Valacyclovir 500 mg orally 2 times/day for 3 days or
Valacyclovir 1 g orally once daily for 5 days
Granuloma Inguinale (Donovanosis)
Azithromycin 1 g orally once weekly or 500 mg daily for >3 weeks and until all lesions have completely healed
Lymphogranuloma Venereum
Doxycycline 100 mg orally 2 times/day for 21 days
Syphillis
Benzathine penicillin G 2.4 million units IM in a single dose
Neurosyphilis, Ocular Syphilis, or Otosyphilis Among Adults
Aqueous crystalline penicillin G 18–24 million units per day, administered as 3–4 million units IV every 4 hours or continuous infusion for 10–14 days
Nongonococcal Urethritis
Doxycycline 100 mg orally 2 times/day for 7 days
Cervicitis*
Doxycycline 100 mg orally 2 times/day for 7 days
- Consider concurrent treatment for gonococcal infection if the patient is at risk for gonorrhea or lives in a community where the prevalence of gonorrhea is high (see Gonococcal Infections).
Chlamydial Infection
Doxycycline 100 mg orally 2 times/day for 7 days
Uncomplicated Gonococcal Infection of the Cervix, Urethra, or Rectum
Ceftriaxone 500 mg* IM in a single dose for persons weighing <150 kg OR
if Ceftriaxone Is Not Available Gentamicin 240 mg IM in a single dose plus Azithromycin 2 g orally in a single dose or
Cefixime* 800 mg orally in a single dose
Uncomplicated Gonococcal Infection of the Pharynx
Ceftriaxone 1 g IM in a single dose
Gonococcal-Related Arthritis and Arthritis-Dermatitis Syndrome
Ceftriaxone 1 g IM or IV every 24 hours
Gonococcal Meningitis and Endocarditis
Recommended Regimen for Gonococcal Meningitis and Endocarditis
Ceftriaxone 1–2 g IV every 24 hours
If macrolide sensitive: Doxycycline 100 mg orally 2 times/day for 7 days, followed by azithromycin 1 g orally initial dose, followed by 500 mg orally once daily for 3 additional days (2.5 g total)
If macrolide resistant: Doxycycline 100 mg orally 2 times/day for 7 days followed by moxifloxacin 400 mg orally once daily for 7 days
Bacterial Vaginosis
Metronidazole 500 mg orally 2 times/day for 7 days or
Metronidazole gel 0.75% one full applicator (5 g) intravaginally, once daily for 5 days or
Clindamycin cream 2% one full applicator (5 g) intravaginally at bedtime for 7 days
Trichomoniasis Among Women
Metronidazole 500 mg orally 2 times/day for 7 days
Trichomoniasis Among Men
Metronidazole 2 g orally in a single dose
Vulvovaginal Candidiasis
Over-the-Counter Intravaginal Agents
Clotrimazole 1% cream 5 g intravaginally daily for 7–14 days or
Clotrimazole 2% cream 5 g intravaginally daily for 3 days or
Miconazole 2% cream 5 g intravaginally daily for 7 days or
Miconazole 4% cream 5 g intravaginally daily for 3 days or
Miconazole 100 mg vaginal suppository one suppository daily for 7 days or
Miconazole 200 mg vaginal suppository one suppository for 3 days or
Miconazole 1,200 mg vaginal suppository one suppository for 1 day or
Tioconazole 6.5% ointment 5 g intravaginally in a single application or
Butoconazole 2% cream (single-dose bioadhesive product) 5 g intravaginally in a single application or
Terconazole 0.4% cream 5 g intravaginally daily for 7 days or
Terconazole 0.8% cream 5 g intravaginally daily for 3 days or
Terconazole 80 mg vaginal suppository one suppository daily for 3 days or
Fluconazole 150 mg orally in a single dose
Pelvic Inflammatory Disease (IV)
Ceftriaxone 1 g IV every 24 hours plus Doxycycline 100 mg orally or IV every 12 hours
plus Metronidazole 500 mg orally or IV every 12 hours or
Cefotetan 2 g IV every 12 hours plus Doxycycline 100 mg orally or IV every 12 hours or
Cefoxitin 2 g IV every 6 hours plus Doxycycline 100 mg orally or IV every 12 hours
Pelvic Inflammatory Disease (oral or IM)
Ceftriaxone 500 mg* IM in a single dose plus Doxycycline 100 mg orally 2 times/day for 14 days with metronidazole 500 mg orally 2 times/day for 14 days or
Cefoxitin 2 g IM in a single dose and probenecid 1 g orally administered concurrently in a single dose plus Doxycycline 100 mg orally 2 times/day for 14 days with metronidazole 500 mg orally 2 times/day for 14 days or
Other parenteral third-generation cephalosporin (e.g., ceftizoxime or cefotaxime)
plus Doxycycline 100 mg orally 2 times/day for 14 days with metronidazole 500 mg orally 2 times/day for 14 days
Epididymitis
Ceftriaxone 500 mg* IM in a single dose plus Doxycycline 100 mg orally 2 times/day for 10 days
External Anogenital Warts (i.e., Penis, Groin, Scrotum, Vulva, Perineum, External Anus, or Perianus)
Patient-applied: Imiquimod 3.75% or 5% cream or
Podofilox 0.5% solution or gel or
Sinecatechins 15% ointment† or
Provider-administered: Cryotherapy with liquid nitrogen or cryoprobe or Surgical removal by tangential scissor excision, tangential shave excision, curettage, laser, or electrosurgery or Trichloroacetic acid (TCA) or bichloroacetic acid (BCA) 80%–90% solution